
This activity can stand alone or be used with the attached worksheet and/or create other worksheets when discussing atom radius or ion radius. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. 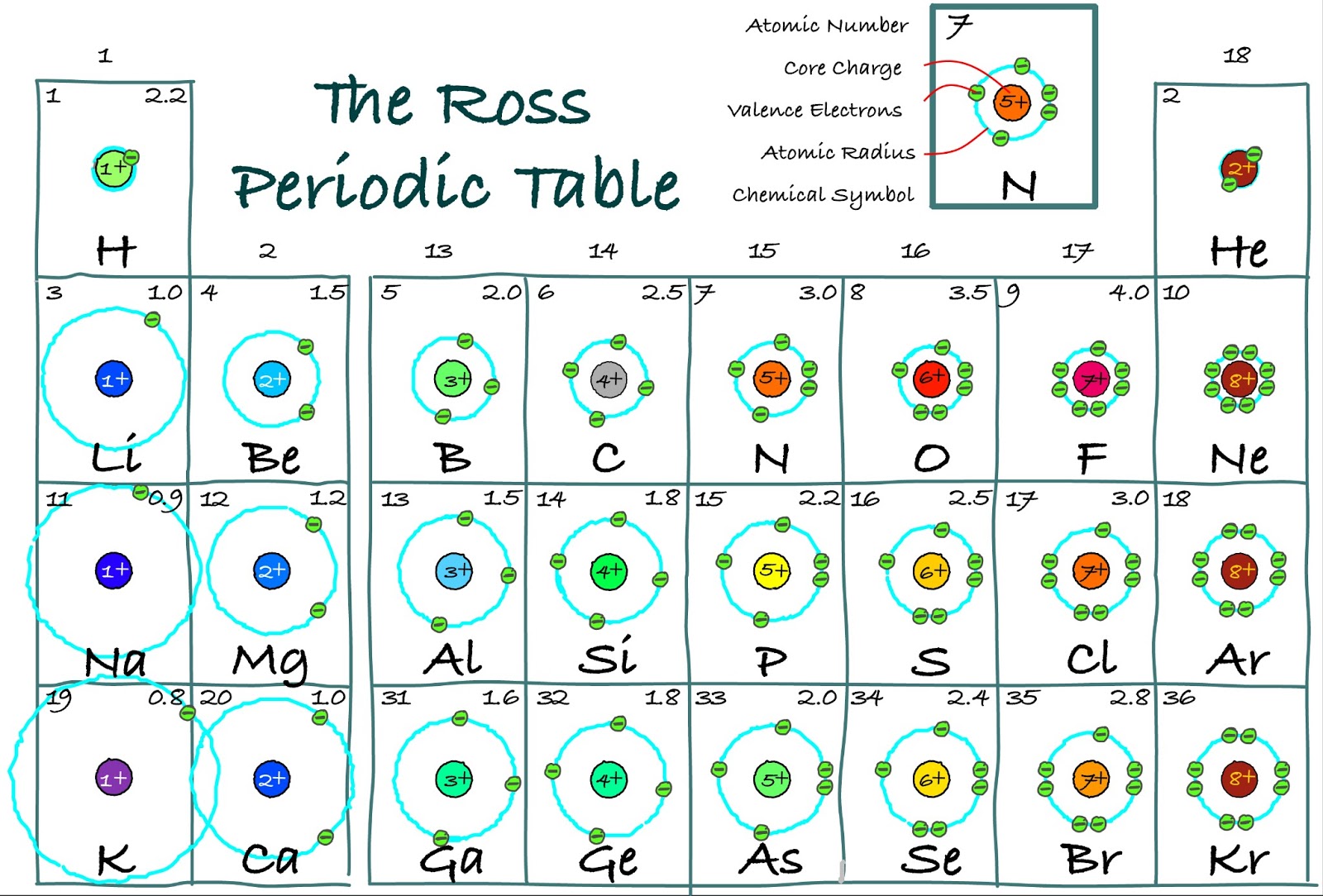
It helps them learn it better than reading it off another chart and it gives them ownership so they keep them longer. Have students add information to the back of their tables like polyatomic ions, equations, key concepts, exceptions to rules. Have students reference their tables when introducing concepts like atomic and ionic radius trends, ionic and covalent bonding regions. This works best when either all the information is on the table or when discussing properties. Information that could be included, but is not limited to, are element name, atomic numbers, mass numbers or weights, common oxidation numbers.Īt some point the columns could be lightly shaded and a legend could be added. This activity has students place the electron-dot model in each appropriate box, but if boxes are large enough other information maybe added throughout the year. Make sure students are filling in the boxes correctly from the start! Aluminum also presents in some gemstones like topaz, garnet, and chrysoberyl. The melting point of the aluminum is 660 ☌ and the boiling point is 2,467 ☌. The atomic weight of the aluminum is 26.9815. Have students keep their tables for a reference tool. Its compound is mostly present in the form of rocks, vegetation, and animals. How many valence electrons does an element have You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. The valence electrons are symbolized as a dot and placed in increasing number around the symbol.Įven though the numbering scheme is no longer widely used it will be helpful later when introducing the idea of half-filled S and P sub-orbital's. On the other hand, nitrogen can form NH 3 so it has a valence of 3, and 3 valence electrons. Where X is the symbol of the element and accounts for the nucleus and all the filled inner shell electrons. In a similar way, the diagram also provides the presentation of tellurium chemical bonding. It shows the numbers of tellurium valence electrons of atoms. It is very helpful to model a couple of electron-dot examples to get students started. The dot diagram helps in the better representation of Te valence electrons. Adding the atomic numbers in the lower corner allows for the addition of mass numbers or atomic weights later if desired. The electron configuration of sodium shows that the last shell. The total number of electrons in a valence shell is called a valence electron. The last shell after the electron configuration is called the valence shell. The third step is to diagnose the valence shell (orbit). It maybe helpful to have students number the boxes one through twenty (atomic numbers) in the lower left corner first to avoid confusion. Step-3: Determine the valence shell and calculate the total electrons. An atoms valence electrons are the electrons in its outermost shell. In this short activity (20-25 minutes) students will be provided a blank periodic table and will fill in the electron-dot model for the first twenty elements. Learn how to determine the number of valence electrons for an element using the periodic table. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.This activity allows students to make a tool of models to help them visualize concepts of the periodic table. The distance between the radii is 228 p m. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. The distance between the radii is 198 p m. Comment déterminer le nombre d’électrons de valence des principaux groupes déléments du tableau périodique et comment représenter leur structure de Lewis à partir de leur configuration électronique. The second diatomic molecule is in a darker shade of green. Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. \): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
